EC stands for electrical conductivity, which is the potential of any material to conduct electricity. Although most growers are used to measuring the amount of feed that they give in ounces per gallon, grams per litre, or some other unit of measurement, EC goes a little further than this. It is important for growers to have a good understanding of what EC is all about and why it matters.
Electrical Conductivity
An EC meter measures the potential for an electrical current to be transported through water. This is known as molar conductivity (electrolytic conductivity) and is measured in siemens (S). Electrons are able to flow through the water from one set of electrodes to another not because of the water molecules themselves, but because of the ions dissolved in the water. It is these ions that transport the electrons.
By the same token, the concentration of ions in the water also determines the number of electrons that can travel from one electrode to the other: the higher the concentration of ions, the greater the flow of electrons. Pure water is a very poor conductor of electricity, which is why an EC meter will read 0.0 in rainwater, reverse osmosis water or de-mineralized water. Salty seawater, on the other hand, is a much better conductor.
When we add nutrients (salts) to water, we increase the molar conductive potential for current through water and thus increase the EC value (or CF = EC*10). All conductivity measurements are directly affected by temperature and this must be allowed for when making them.
EC units
Electrical conductivity can be expressed using a number of different units, but the typical unit is siemens per meter2 per mole (S/m2/mole) or millisiemens per centimetre (mS/cm). The mS/cm unit is generally used in Europe as a guide to the concentration of nutrients in water. In North America, conductivity is converted into a count of the ions in the water using parts per million (which can also be converted into units including mg/l etc.). This is done by converting the EC into a value based on the ions contained in the solution. Fortunately, there is a fixed calculation for the relationship between all these units, which is given in the table below.
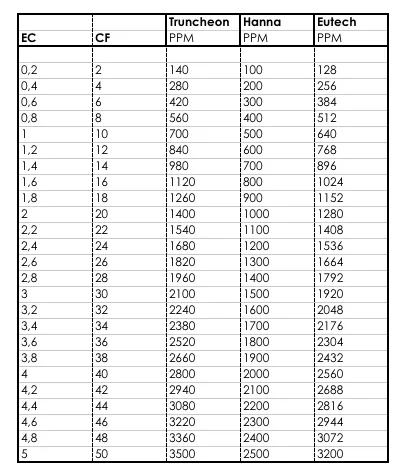
Is an EC value nutritional?
Water that contains mineral salts has an EC but the presence of EC alone does not necessarily indicate that the water contains nutritional salts that will help plants. Tap water can contain sodium and chloride, for example, which have an EC value but no nutritional value for plants.
Fertiliser is made up of nutritional salts, of course. Any nutritional value that we add to water is known as EC+ and should be added to the water’s residual EC. This is how we measure the total EC in our feeding tank.
Producing a high EC
Salt has the property of making the water attracted to itself, a process known as hydrolysis. A pot of salt placed in a cellar will reduce the atmospheric humidity, for example, by attracting water from the atmosphere. In a solution, the concentration of salts will always try to equalize between two areas with different concentrations – in other words, the water will move to the area of higher concentration. This difference in concentrations is known as the water potential gradient, and it also plays a role in our cultivation through a process known as osmosis.
Osmosis involves a semi-permeable barrier that allows water molecules to pass through but restricts the movement of the ions or salts in solution. When we dissolve many nutrients in water (producing a high EC), the nutritional salts attract the water in the substrate to themselves. This makes it more difficult for the roots to extract the water from the substrate. So it is actually possible for us to create conditions where the roots are no longer capable of extracting any more water from the substrate, even though the substrate is saturated. This is known as making the substrate 'physiologically dry'. The result is that no more water is available for the plants to cool themselves through transpiration (evaporation), which they need to do when heat and light are present.
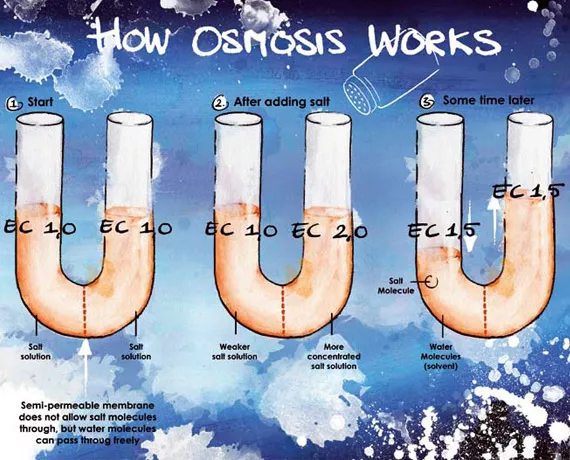
We can see this osmotic process in action a U-tube if we separate the two sides with a permeable membrane (such as a piece of stem). If we now add some salt to one side of the tube, the water level on that side will rise because the water with a lower EC (a lower concentration of salts) will be drawn towards it (see fig. 1). All this means that it is important to add little or no nutrients at the very start of the growing process.
Internal EC
Once the plant has taken up nutritional material from the feed solution, we need to try to build up the plant’s osmotic value (or its internal concentration of salts) as quickly as possible. Because the volume of the plant increases as it grows and absorbs water, the osmotic value falls. The salts within the plant redistribute themselves and the plant becomes softer and lighter coloured. This makes it very susceptible to dehydration (wilting) because water can leave the plant easily.
Giving more nutrition to the roots will show proportionally in the growth. Because the water that is used to transport the nutritional salts has evaporated, the salts will remain in the plant, raising its internal EC (osmotic value). This means that the grower can expose the roots to a solution with a higher EC again.
EC build-up
By achieving this positive spiral of EC build-up in the plant, the plant also becomes more capable of absorbing and retaining water. This means that water does not evaporate from the plant too easily and it will not dehydrate too quickly. The table below shows an example of a plant that has lost its water reserves too soon.

When plants become too soft, the intensity of the light must be reduced or the number of hours of lighting shortened to prevent them drying out at the end of the day. Even though EC plays an important role in this story, it is not the only factor that has an influence. The overall climate around the plant influences the processes of which EC is a part.
Nutritional needs
When building up the plant’s internal EC and subsequently that of the substrate, it is important to take into account the demands of the plant’s growth. This demand is controlled by assimilation. The bigger a plant grows, the more nutrition it will need. These nutrients are partly locked up in the plant and converted into amino acids, oils, fats etc., but some nutrient salts also remain in the plant’s sap and these determine the plant’s internal EC. Potassium is one of the most important nutritional elements for this.
Once the plant has finished the vegetative growth phase, it can still absorb a lot of potassium for its internal osmotic value and the ovaries. The ovaries are not the fertilised 'seed'. However, this increasing rate of uptake comes to an end. After approximately 60% of the cultivation cycle, the plant will have taken up enough nutrients from the stock in the substrate. The game of nutrient stock versus applied EC now starts for growers.
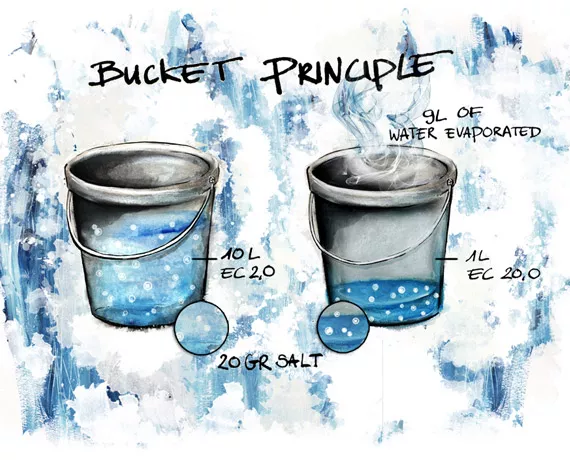
EC stock in the pot
We can use the “bucket” principle to understand this game.
Summary
As well as being a unit for measuring the fertiliser given to plants, EC is also a climate control mechanism that relates to water absorption.
Plants should start growing with a low EC, and this should then be built up as quickly as possible in order to provide for the plant’s nutritional requirements as well as to raise the internal osmotic value to build a stronger plant.
The plant requires hardly any further nutrition in the final weeks of cultivation. We only continue to provide nutrition in order to maintain the nutritional stock in the pot and keep the EC stable. This generally results in the EC being lowered or even in a weekly rinse (leaching).




