All over the world, there is debate about the exact definition of hard water and soft water, and how this should be taken into account when feeding plants. The hardness of water depends on the concentration of positive metallic ions dissolved in the water, typically calcium and magnesium.
This effect can be heightened by the presence of dissolved carbon dioxide (CO2) in the water as carbonates or bicarbonates. ‘Soft water’ can be defined as 60mg/l, and water with over 120mg/l is considered to be ‘hard’, while water in between is defined as moderately hard. However, different agencies have their own definitions (see table 1).
Hard water, soft water, bad water... what’s the difference? Anything dissolved in water can and will react with other elements that are added to the water, or with anything that the water comes into contact with. Hard water is an issue for cleaning and equipment, and it increases the chemical reactiveness of the water especially as far as pH is concerned; it is often considered healthier. Typically, hard water comes from ground water that has been exposed for longer periods to mineral-bearing rocks. Well water is a prime example.
Soft water on the other hand allows soap to foam up and work better. It affects equipment less, and provides more of a ‘blank slate’ for chemical reactions. Studies have shown a correlation between soft water and health issues including cardiac disease. Soft water is typically sourced from surface water − rivers, streams, and lakes − and has not been exposed for long periods to mineral bearing rock formations. It can also come from treated water, where most ions have been removed or replaced with single valance atoms such as sodium by water softening equipment.
TDS, EC and water hardness
It’s important not to confuse ion or salt concentration with the hardness or softness of water. Hardness is a function of multivalent ions such as Ca2+ and Mg2+, not monovalent ions like Na+ or Cl+. Monovalent ions also show up in the total dissolved solids (TDS) of a solution, so it is possible to have a TDS of 450 mg/L (1 ppm = 1 mg/L), derived from adding table salt to distilled water, but this water can still be soft.
There is no direct correlation between TDS or electrical conductivity (EC) and water hardness. You can only be sure that the water is hard if you can know that all the electrical conductivity derives exclusively from Ca, Mg or other multivalent metallic ions. For example, sugar solution will conduct electricity, but is not (necessarily) hard. Water softeners work by replacing the problem ions − calcium and magnesium − with sodium ions. The EC stays the same or increases but the water goes from hard to soft; not a good thing for plants.
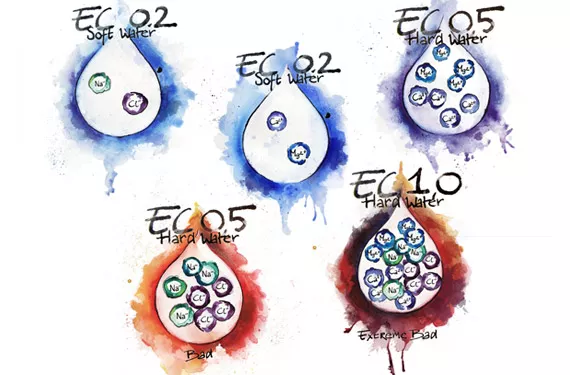
For us, the big question is how all this affects plant nutrition. One of the biggest effects for growing systems that use hard water is the potential for a build-up of calcium carbonate or magnesium carbonate, which are insoluble. The formation of these deposits is an endothermic reaction, meaning that the process gets faster as heat is supplied to the solution. During the process of pumping water from a reservoir, through a pump, through smaller pipes, onto a table top and through a root system, the water will become warmer, and this will cause the deposits to form naturally and persistently.
Relationship between elements
As the heat enters the system, the formation of calcium or magnesium carbonate increases, resulting in the deposition of insoluble materials on the inside of pumps, pipes, tubes and the medium of the growing system. Ultimately this leads to restricted flows, blocked emitters, burnt-out pumps, and so on.
The effect of the nutrient package on the chemical profile of the water can also be altered by various antagonistic relationships between individual elements and the overall effect on pH. The harder the water, the more calcium and magnesium is available. The more of these elements there is in relation to certain other elements, such as potassium and phosphorous, the less available those elements become, so they are effectively locked out. These positive ions will raise the pH of the solution, and when hardness is also affected by carbonate levels, the pH effect will continue into the medium which it is applied to. The harder the water, the more acid is required to lower the pH.
Table 1.
Recommended upper limits of chemical factors in irrigation water for greenhouse crop production (Based on 1 and 2, see bibliography)

Moderate levels of hardness
There are a number of commercial solutions to moderate levels of hardness:
1. Water softening. Water softening involves flooding the water with a monovalent ion, typically Sodium, which drives out the Calcium and lowers the hardness of the water. This is great for clothes washing and baths but not so great for water that you are using to supply your plants or for human consumption, especially where the water is very hard to start with.
2. Reverse osmosis (RO), a process where tap water is forced through a series of membranes with progressively smaller pores which block molecules and atoms over a certain size. This filters out the calcium and other larger elements effectively lowering the hardness of the water. It also strips out most other elements including harmful molecules, sodium ions, and most other ions thus effectively lowering overall total dissolved solids and EC. However, it is also expensive to install and maintain and not always completely necessary − at least, using pure reverse osmosis water is unnecessary.
Water and growing systems
Only soft water is recommended for recirculation, and only when an inert growing medium is used. This can be done with pure reverse osmosis water, for example. Recirculating systems must be able to adjust not only to the hardness of the water, but also to the additional elements supplied in the tap water over and above what is added or needed in the nutrients added. Controlling salt composition is critical because this also affects pH levels, which are critical in signaling a plant’s flowering response (in addition to photoperiod change). It is best to use a nutrient which is designed to work with tap water with EC values no greater than 0.3 – 0.4 mS/cm while providing some buffering for pH control in the system (for example CANNA AQUA).
Another method used in growing systems is the run-to- waste system where tank-mixed nutrients are applied to the plants and the excess is allowed to drain away and not re- captured. In this system, it is important to not only regulate the pH when mixing it, but also to maintain the pH over time while the prepared product is stored in the tank before use. This minimizes pH swings while preventing insoluble compounds from forming. Also, there are less calcium and magnesium ions available in soft water, so those need to be augmented or replaced to achieve the correct ratio of ions. If you want to avoid worrying about nutrient composition, you should choose a nutrient that has both a soft water and hard water versions (for RTW) (like CANNA SUBSTRA). How do you know whether to use the hard water or soft water version? Simple: run a water sample and compare the results with the table above.








